 
Polar Bonds - In Polar Covalent chemical bonding, electrons are shared unequally since the more electronegative atom pulls the electron pair closer to itself and away from the less electronegative atom.Chapter 9 Review Video Answers Videos on Hybridization and sigma/pi bonds. Hydrogen Bonds - It is a type of polar covalent bonding between oxygen and hydrogen wherein the hydrogen develops a partial positive charge Bonds and Molecular Structure 1) How are ionic bonds and covalent bonds.Covalent Bonds - Compounds that contain carbon commonly exhibit this type of chemical bonding.Ionic Bonds - Ionic bonding is a type of chemical bonding which involves a transfer of electrons from one atom or molecule to another.So, the two carbons in ethylene, which is the first member of the. In sigma bonds, orbitals may overlap: two hybrid orbitals, one hybrid and one pure orbital or two pure orbitals. Pi bonds are formed through the lateral overlap of the half - filled atomic orbitals. Sigma and pi bonds are types of covalent bonds that differ in the overlapping of atomic orbitals. 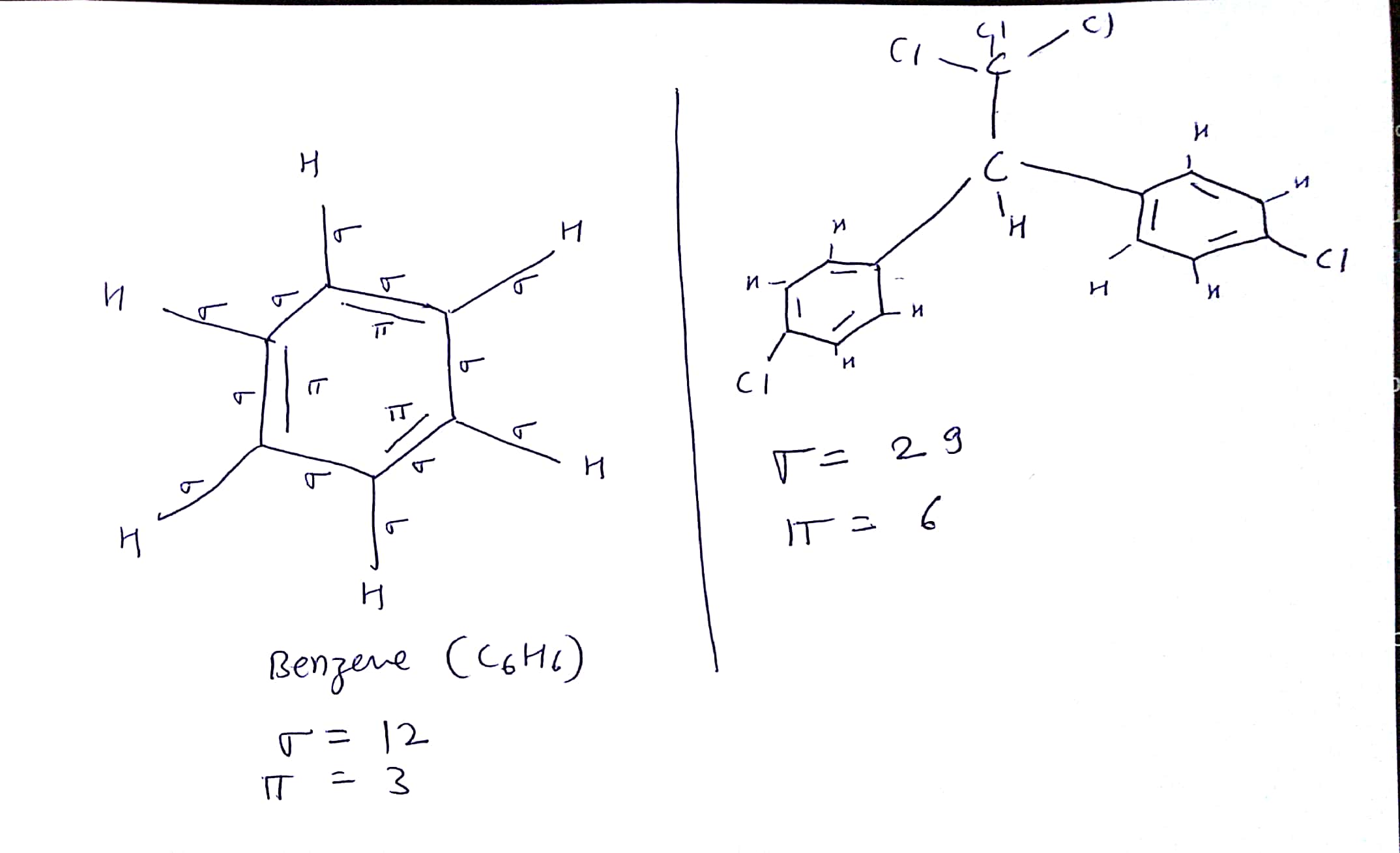
The two carbons are sp2 -hybridized where the sp2 orbitals overlap to form the sigma bond, and the unhybridized p orbitals form the pi bond. Sigma bonds are formed by the axial overlap of half- filled atomic orbitals. 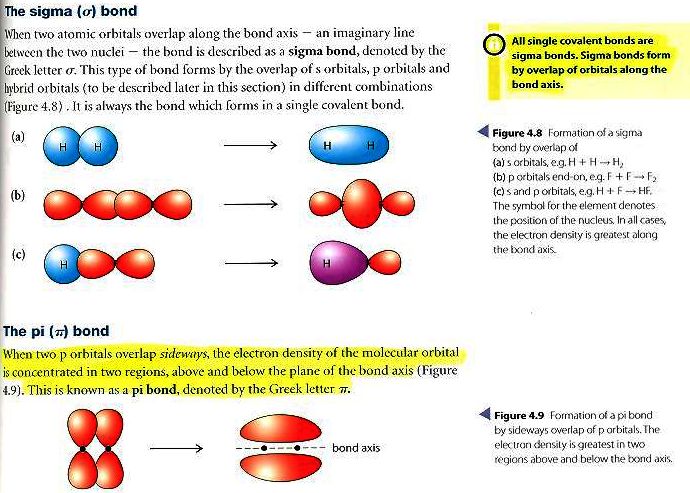
There are 4 types of chemical bonds which are formed by atoms or molecules to yield compounds. The pi bond is formed by a side-to-side overlap of two p orbitals provided by adjacent atoms. The attractive force which holds various constituents (atoms, ions, etc.) together in different chemical species is called a chemical bond. Obviously, there must be some force that holds these constituent atoms together in the molecules. Such a group of atoms is called a molecule. We have 25 more practice problems on Lewis Dot Structures: Sigma & Pi Bonds. This chemistry video tutorial provides a basic introduction into sigma and pi bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
